
Explore the now-ruined estates of the Irish countryside and you occasionally find a stone cylinder, as much as several metres high and wide, open at the top and with a small door at the base.
Some resemble the medieval fortresses that still dot the landscape here – but no one built fortresses so tiny, or half-buried in the side of a hill. In fact, they are kilns for lime burning, a now-forgotten industry that sustained many agrarian communities before energy became cheap.
Limestone is mainly coral and shells of long-extinct sea creatures, squeezed over aeons into a solid mass of calcium carbonate
“Lime” here means neither the citrus fruit nor the tree, but refers to a white powder derived from limestone. For at least 7,000 years humans created lime in kilns, as they might have hardened pottery or smelted ore, and used the material for dozens of purposes now largely replaced by fossil-fuel by-products – perhaps most commonly to create mortar for construction.
British and Irish farmers, though, found it most important to neutralise acid soils and multiply crop production – as much as fourfold, by some contemporary accounts. For hundreds of years until the mid-20th century, lime supported a vast and vital network of village industry —
quarries to mine the limestone, carts and barges to transport it, and specialists to monitor the burning. In the late 1700s, according to one survey, County Cork alone was said to contain an amazing 23,000 kilns, or one every 80 acres. 1
Limestone is mainly coral and shells of long-extinct sea creatures, squeezed over aeons into a solid mass of calcium carbonate, or CaCO3. When burned at 900 degrees C or more it vents carbon dioxide (CO2), leaving behind the volatile calcium oxide (CaO) – “quicklime,” “burnt lime” or “unslaked lime.”
Then, when combined with water – hydrated or “slaked” — the quicklime became calcium hydroxide or Ca(OH)2, and could be put to many uses. Confusingly, all of these have been called “lime” at times, but in this article, we will call the original rock “limestone,” the caustic material from the kiln “quicklime,” and the hydrated final product “lime” for clarity.
Roman Concrete
The earliest use of lime dates to present-day Turkey between 7,000 and 14,000 years ago, and many ancient civilisations used it to create mortar between stones. The Romans, however, took lime a step further, mixing it with various other ingredients to create an early version of cement. In fact, their version has proven superior to our own in some ways. Our concrete lasts only decades – as little as a single decade in seawater — while Romans created concrete that not only formed in seawater, but have withstood the pounding of waves for 2,000 years.

The secret, according to two papers released in the summer of 2013, involved mixing quicklime with volcanic ash to form mortar. Volcanic ash was plentifully gathered from the volcano at Vesuvius, according to Pliny the Elder – ironically, the same volcano that would later kill him. Romans then packed this mortar into wooden forms and lowered them into seawater, which caused the quicklime to react and form a lime-and-ash mix of waterproof cement.
The papers’ authors say such techniques could prove useful even today; not only did their concretes stand up to time and the elements better than ours, but such methods are “greener” – generating less carbon emission – than our cement manufacture. Crushing rocks into Portland cement powder requires enormous quantities of energy and accounts for seven per cent of all industrial carbon emissions on the planet. 23456

Romans brought such technologies with them as they spread across Europe, so lime kilns appeared in Britain with their invasion and disappeared for several hundred years after they left. In Ireland, where Romans never set foot, Normans apparently brought the technology in the 1200s, to build the round towers that still frequently stand today. (7)
Whitewash, Limelight and other Applications
Lime also forms the basis of whitewash, used for centuries to protect and brighten structures, fences, vehicles and even trees, without the alarming and unpronounceable stew of toxic ingredients in many modern paints. Whitewash is fundamentally a mix of lime and water, although it could also contain salt, milk, linseed oil for water-proofing, or hair or cereal husks for strength.
The dried lime was safe to handle and even for animals to lick, but remained mildly alkaline enough to disinfect barn and dairy walls. Its brilliant whiteness was valued in places like Britain and Ireland, where the winters grow very dark – Irish cottages were traditionally whitewashed in spring and again before Christmas. In sunnier climates, however, that same colour helped keep buildings cool.

Lime had many other uses: Farmers rubbed it on their livestock’s feet as an antiseptic, or painted it onto fruit trees to prevent fungal diseases. Some mixed a bit of lime into well-water to disinfect it, or to preserve eggs for months without spoiling. Tanners used it to remove hair from hides, gardeners to repel slugs and snails, printers to bleach paper.
Even the corrosive quicklime, the calcium oxide that came straight from the kiln, had many uses before it was hydrated. It kept pantries and store-rooms dry – the 1915 household manual “The Best Way” recommended keeping a bowl of it to reduce humidity, as it sucked moisture from the air. It caught fire easily – sometimes too easily – and was used to make an early, high-intensity lamp for the stage – the original limelight. 7
Terrorist Weapon
It also made a rather fearsome weapon, as it could sear the skin and blind the eyes. In David Hume’s A History of England, he recounts a battle between English and French ships around 1216, in which the English captain Phillip d’Albiney ingeniously used quicklime to turn the tide of battle. He saw that the winds were blowing from his ships to French fleet, and “having gained the wind of the French, he came down upon them with violence; and throwing in their faces a great quantity of quick lime, which he purposely carried on board, he so blinded them, that they were disabled from defending themselves.”

The compound made a handy terrorist weapon as well; when Irish reformer Charles Parnell spoke at a political rally in 1891, someone in the crowd threw quicklime at his face, and “had not [he] shut his eyes in time, he would undoubtedly have been blinded,” his wife Katherine later wrote.
Quicklime was also shovelled into graves to decompose bodies more quickly, as Oscar Wilde saw when he was a prisoner at Reading Gaol (Jail) in Britain:
And all the while the burning lime Eats flesh and bone away
It eats the brittle bone by night
And the soft flesh by the day
It eats the flesh and bone by turns
But eats the heart away.
Lime in Agriculture: Sweetening the Soil
Its use in agriculture, however, eclipsed any other use on these islands, so valuable was its ability to turn acid bog-lands into croplands. Some 40 per cent of the arable land in the world is too acidic for many plants to grow – the more acidic the soil, the more toxic aluminium plants absorb. These days, farmers often treat such soils with crushed limestone or other energy-intensive products, and scientists like Chris Gustafson of the University of Missouri are trying to genetically engineer aluminium-resistant crops. In earlier eras, however, farmers found that lime temporarily “sweetened” or neutralised the soil. 8
This made lime so valuable that many agrarian communities supported a network of local industries to create it — quarries to mine the limestone, wagons to transport the rocks by road or barges by canal, and specialists to supervise the burning. By the mid-1600s many families in County Cork, Ireland, for example, paid their rent by lime-burning on the side, according to a civil survey of the time. 9
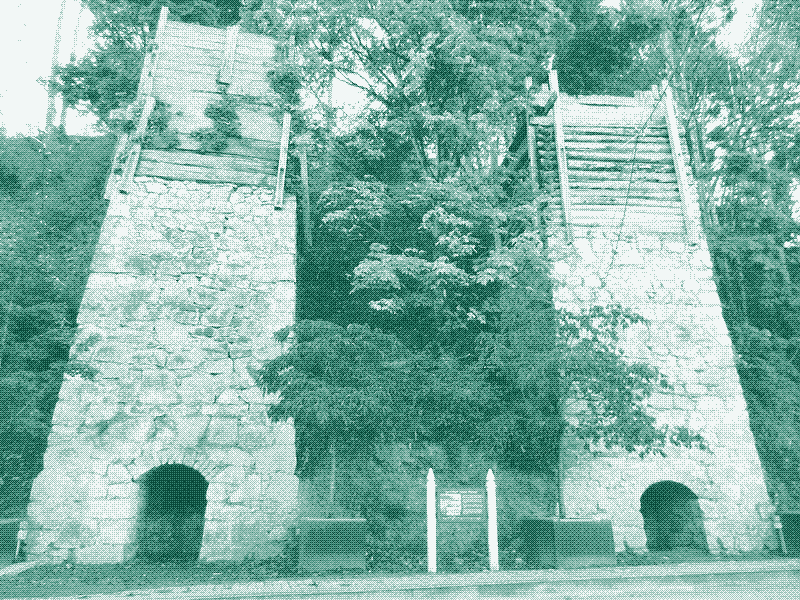
Farmers treated the soil in quite a straightforward manner: they shovelled quicklime straight from the kiln onto a horse-drawn cart, drove the cart to the needed field and drove the horse back and forth across it as though ploughing. Every several metres the farmer stopped the cart and scooped several shovels of quicklime in “falls” on the ground — six to eight barrels to the acre.
Spreading a highly caustic compound onto cropland might sound inadvisable, but the next rain both hydrated it into lime and soaked it into the ground. Transporting the quicklime, however, was dangerous work, as it could spontaneously burst into flame and burn carts and barns, or simply to eat through wooden containers if it wasn’t spread quickly. 1011
The process only sweetened the land for a limited amount of time, according to contemporary reports – three years in some fields, twelve years in others, depending on conditions. In any case liming had to be continually re-applied or it “enriched the father but impoverished the son,” went the saying, so the kilns were kept in steady business. 12
Operating the Kiln
Kilns themselves needed to be carefully situated: they needed to be as close as possible to quarries, so that hundreds of tonnes of rock could be carried with as little effort as possible, by horse or barge. At the same time they had to lie as close to the lime’s destination as possible – a fortress or church being built with mortar, or fields that needed sweetening — so that the quicklime could also be transported without incident. Moreover, they could not be situated near populated areas or even campsites, as the burning lime gave off noxious and potentially lethal gases.

The brick or stone structures were often built into hillsides to allow people to easily transport coal and lime to the open top, or mouth, and were often several metres across and about as high. On the inside they usually tapered down so that gravity alone fed the fuel down, and at the narrow bottom of the cone, one wall had an arched opening or “eye.”
The kiln had to be filled carefully, with precisely measured amounts and materials – if the lime did not bake at a high enough temperature for long enough, the stone would not transform into quicklime and the work would be in vain. Lime-burners filled the bottom of the kiln with the driest wood possible – furze-wood was often mentioned – and then the men lay alternating layers of fuel and limestone.
Perhaps the most common fuel was “colm” – anthracite coal – although charcoal could also be used, as well as “turf” – dried peat from the bogs here. Whatever the fuel, it had to be in an opaque layer, insulating the chunks of limestone from the sides of the kiln and from each other, according to old lime-burners interviewed decades later for Irish national radio.
Sleeping by the Kiln
Once the kiln was filled, the wood – at the bottom of the kiln, by that little door – was set on fire, and that, in turn, lit the fuel through the rest of the structure. Once the kiln was lit there was no going back; the lime-burners had to maintain a watch over the kiln for the next three or four days, sleeping nearby.
Burning was often done in winter, when there were fewer farm chores to be done, so it must have been tempting for men sleeping out in the cold to move closer to the warm glow of the kiln. According to lime expert Colin Richards, however, sleeping by the kiln was extremely dangerous, between the poison gases and the open pit. There were cases of itinerants sleeping near the mouth for warmth, he said, rolling into it as they slept and being roasted alive.

Certainly the men did exhausting work for days at a stretch, making them “thirsty as a lime-burner” as the saying went. A single kiln could hold a hundred tonnes of material, which had to be shovelled in by hand, yet delicately measured and arranged inside. Of course there was less to shovel out – the coal had burned away, and the limestone had lost some of its mass – but that material was much more difficult to handle.
“Drawing out the lime underneath was the dirtiest part of it,” said one anonymous lime-burner who worked in Ireland in the 1930s and 40s and was interviewed for a radio documentary in 1981. “It was there that you got the dust, and you got too much of it and you began bleeding from the nostrils.”
Magic and Ritual
With their furnace-like heat, poison vapours, alchemical transformations, hazardous products and vital importance to agrarian survival, it was perhaps inevitable that farmers associated kilns with all kinds of magic and ritual. According to Irish elders interviewed in the 1930s, young people often performed Halloween rituals around burning-the-bones-of-the-earth-lime-kilns to find out who they would marry.
In one instance, fairies were said to have killed off a farmer’s livestock after he inadvertently built a kiln in their way. Other peoples were said to have summoned evil spirits there; a reverend in Carnmoney, rumoured to have sold his soul to the Devil, was said to have courteously invited him to a kiln so the Devil would feel at home. (15)(16)(17) The lime burners themselves had a simpler ritual, one they said was practiced among “all the lime burners of old.”
“You took a bottle with you that morning … of holy water,” one said, and before the kiln was fired up “you just sprinkled it on top the stones, and made the Sign of the Cross, for you were burning – what they used to say was — you were burning the bones of the Earth.”
Brian Kaller is a journalist living in rural Ireland. He blogs at Restoring Mayberry
Reactions
To make a comment, please send an e-mail to solar (at) lowtechmagazine (dot) com. Your e-mail address is not used for other purposes, and will be deleted after the comment is published. If you don’t want your real name to be published, sign the e-mail with the name you want to appear.
Reactions
Kelly
Very interesting article, I’ve always wondered about the various limekilns we see just about everywhere, and what their purpose and history is.
One minor correction: you say of quicklime, “It caught fire easily – sometimes too easily – and was used to make an early, high-intensity lamp for the stage – the original limelight.”
Of course, quicklime is not flammable - if it were, it would be consumed in the kiln! A ’limelight’ is created by heating calcium oxide to a very high temperature - via an external heat source, such as a flame - until the calcium oxide gives off bright light via incandescence. The quicklime does not - and cannot - burn.
Joel
Quicklime is non-flammable, but there is yet another substance named “lime” that this article didn’t mention.
Over-burned lime (calcium carbide) releases acetylene when it gets wet. Acetylene is very flammable, and a lot of heat is evolved when lime is “slaked”, so I could imagine a wagonload of lime catching fire spontaneously, if the limeburners had originally used too much fuel and conditions were humid.
This method of making acetylene is how blowtorches were fueled; the evolving gas would push down the level of water in the container, regulating the pressure automatically using gravity. It’s a neat system, if not particularly efficient.
Matthas
The CO2 emissions from the cement industry are not becaue they use so much power for the milling. It is the CO2 which is set free in the precalciner, exactly the same process as burning lime. For 1.6 tons of raw meal you get one ton of clinker. The remainder is mostly CO2 which leaves trough the stack. A cement plant which produces 10000 tons of clinker a day will release 6000 tons of CO2 . Depending on the fuel it will a bit more. The emissions caused energy consumption of the mills are marginal if you compare to this numbers.
Cement plants nowadays use large amount of secondary fuel. The burn all kinds of waste. As the temperature are so high, even some toxic fuel can be used.
By the ton modern cement plants are more energy efficient and the gas can be cleaned in several stages. First plants will generate electricity out of waste heat.
Also there are many types of cement not only Portland cement. Some have similar properties like the Roman cement.
Michael Gambill
A fascinating look at the historical significance of this technology. Understanding past advancements equips us to make informed decisions about today’s technology. It is a point that I regularly make with my high school history students. Well done and thanks.
Lindsapril
Loved this article. I am currently writing a masters thesis on lime kiln use in early settlement Saskatchewan - it was nice to see a concise and interesting write up on the traditions these homesteaders in Saskatchewan were drawing their knowledge and skill from. Thank you!
Himbeerkuchen
Thanks a lot for the article. This is a very interesting one, as it clearly shows that in the ‘good old days’ not everything was sustainable either. Especially note the ’toxic fumes’ bits…
However, the footprint is still different. Just compare those relatively few kilns with the incredible numbers and area of industrial ruins and wasteland that we are leaving to our descendants. A shame.
RobD
Nice article.
As a variation:
In our sea-side village in Holland a kiln (one of many in the past)has remained that used shells as raw material; no limestone in this country.
Peter Dew
Presuming that slaking lime refers to adding water to the quick-lime to make hydrated lime or calcium hydroxide for mortar, is there a specific term for burning the limestone to make un-hydrated or quick-lime ? It doesn’t seem right that they simply burnt or burned limestone, which I understand may have been a key industry based on salvaging it from ruins in Medieval and Renaissance Rome.
Lisa
To Lindsapril, I found your thesis and congratulations on a fine work.
eric koperek
Response to Joel (comment # 2 above): Calcium Carbide = CAC2 is NOT “over baked” lime! Calcium carbide was not invented until 1892. Calcium carbide is made from limestone and coke heated in an electric arc furnace at 2200 degrees Centigrade. These temperatures are NOT possible with common lime kilns or open fires. end comment.
Burned Lime vs. Agricultural Lime: It is not necessary to burn limestone rock in order to “sweeten” agricultural soils. Powdered agricultural limestone, chalk, or crushed shells work just as well and are much less costly. If possible, use dolomite limestone or dolomitic limestone as these rocks also contain substantial amounts of magnesium, a necessary component of plant chlorophyll. Rule-of-Thumb: Most agricultural soils east of the Mississippi River require 2 tons = 4,000 pounds of limestone per acre (1.5 ounces per square foot) to keep a slightly acidic soil pH of 6.5 to 6.8. Slightly acidic soils help plants absorb essential micronutrients. If limestone is not available or too costly, apply common wood ashes or agricultural charcoal at 4,000 pounds per acre (3 ounces per square foot). 2 pounds of wood ash or agricultural charcoal = 1 pound of powdered agricultural limestone. NOTE: Always mix agricultural charcoal with an equal volume of manure and compost for 30 days before application. Do not apply uncomposted charcoal to the soil or it will soak up nitrogen and your crops will be yellow and stunted.
Richard Rhodes
Brian:
Thanks for your impressive work on this technical and somewhat difficult subject. Many people get this wrong and I find your article and research illuminating. I am sure others will value it as well.
Keep up the good work,
Richard Rhodes
Caroline
The first photo in this post, shows a harbour marker to the north of Porthgain harbour, not a limekiln.
Beth Jabornik
A fascinating article. Thank you for publishing this. I am researching kilns in Australia.
Amy Roddey
This is a fascinating piece. I want to read everything else you have written and learn what you know about other subjects as well. Thank you for putting this together and sharing. I have three sons and I think I know what you mean.